Origin of strong CO binding and low C-C coupling barrier for ethanol synthesis on non-metal doped graphyne: Bond order conservation and flexible orbital hybridization
Poobodin Mano, Supawadee Namuangruk, Kaito Takahashi
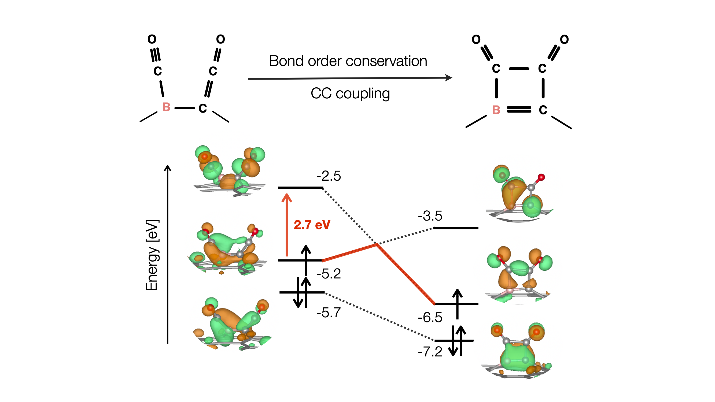
For CO2 electroreduction to produce C2+ products, many non-metal catalyst studies have focused on doping to sp2 and sp3 carbon material for efficient Carbon-Carbon (CC) bond formation. However, studies on doping to sp hybridization are scarce, so we focused on graphyne (GRY), a stable carbon allotrope with sp-sp2 hybridization. We theoretically showed that ethanol could be selectively produced with a low limiting potential of -0.53 V with a very low CC coupling barrier of 0.46 eV by boron doping (B-GRY) to the highly abundant sp hybrid linker. Fascinatingly, strong CO binding energy coexisted with a low CC coupling barrier, thus breaking the Bell-Evans-Polanyi relation. This provides a new direction that differs from previous studies that utilized the weakening of CO binding energy to reduce the CC coupling barrier. Our calculations using an integrated crystal orbital bond index showed that for 2*CO, the binding with B-GRY includes both s donation and π back donation, while only s binding with B-GRY occurs for *COCO. The electrons responsible for the π back donation for CO binding were used for the OC-CO bond formation after CC coupling. The reactive π electron on the sp hybrid linker has a chameleon-like feature changing its orbital hybridization to donate electrons depending on the adsorbate. We utilized the integrated crystal orbital bond index along the ethanol production pathway for the first time and showed that the bond order conservation effectively promotes CC coupling on B-GRY.