Effect of 3d-transition metals doped in ZnO monolayers on the CO2 electrochemical reduction to valuable products: first principles study
Pornsawan Sikam, Kaito Takahashi, Thantip Roongcharoen, Thanadol Jitwatanasirikul, Chirawat Chitpakdee, Kajornsak Faungnawakij, Supawadee Namuangruk*
Applied Surface Science
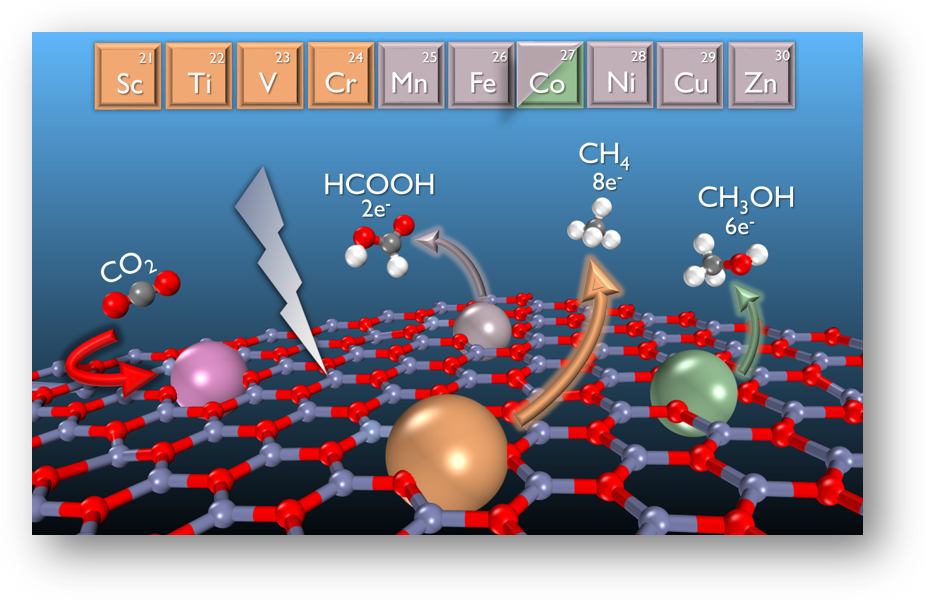
CO2 conversion to valuable products on ZnO (0001) monolayer doped by transition metals (TM-ZnO where TM is Sc, Ti, V, Cr, Mn, Fe, Co, Ni, and Cu) was investigated by density functional theory calculation. The results show that doping TMs can reduce the overpotential for CO2 reduction reaction (CRR) compared to pristine ZnO. Significantly, the oxidation state of TMs by different d-orbital occupancy results in a change of the electronic properties of the catalysts, leading to a difference in reactivity, reaction pathway, and selectivity of the final products. Early TMs (Sc to Cr) showing oxidation state 3+ prefer CH4 as a product while late TMs (Mn to Cu) showing oxidation state 2+ can make HCOOH. Remarkably, Co-ZnO can produce HCOOH with ultra-low overpotential at 0.02 V and can further produce CH3OH with an overpotential of only 0.45 V. Therefore, Co-ZnO monolayer is suggested as a promising CRR catalyst for experimental research. This work sheds light on the rational design of low-cost metal oxides with high stability, activity, and product selectivity for CRR and other reactions.