The study of HIV-1 Vpr-membrane and Vpr-hVDAC-1 interactions by Graphene Field-Effect Transistor Biosensors
Peibin Zhong,# Chun Hao Liu,# Yit-Tsong Chen* and Tsyr-Yan Yu*
ACS Appl. Bio Mater. 2020, 3, 9, 6351–6357; DOI: 10.1021/acsabm.0c00783
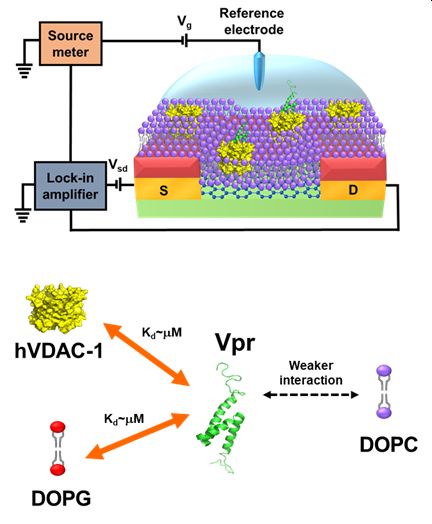
The viral protein R (Vpr) of human immunodeficiency virus 1 (HIV-1) is involved in many cellular processes during the viral life cycle; however, its associated mechanisms remain unclear. Here, we designed an E. coli expression construct to achieve a milligram yield of recombinant Vpr. In addition, we fabricated a graphene field-effect transistor (G-FET) biosensor, with the modification of a supported lipid bilayer (SLB), to study the interaction between Vpr and its interaction partners. The Dirac point of the SLB/G-FET was observed to shift in response to the binding of Vpr to SLB. By fitting the normalized shift of the Dirac point as a function of Vpr concentration to the Langmuir adsorption isotherm equation, we could extract the dissociation constant (Kd) to quantify the Vpr-binding affinity. When 1,2-dioleoyl-sn-glycero-3-phospho-(1'-rac-glycerol) (DOPG) mem-brane was used as the SLB, the dissociation constant was determined to be 9.6 ± 2.1 μM. In contrast, only a slight shift of the Dirac point was observed in response to the addition of Vpr when 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) membrane was used as the SLB. Taking the advantage of the much weaker binding of Vpr to DOPC membrane, we prepared human voltage-dependent anion channel isoform 1 (hVDAC-1) embed-ded DOPC membrane as the SLB for G-FET, and used it to determine the dissociation constant as 5.1 ± 0.9 μM. In summary, using clinically relevant Vpr protein as an example, we demonstrated that a SLB/G-FET bio-sensor is a suitable tool for studying the interaction between a membrane associated protein and its interac-tion partners.