Label-free detection of protein-protein interactions using a calmodulin-modified nanowire transistor
T.-W. Lin, P.-J. Hsieh, C.-L. Lin, Y.-Y. Fang, J.-X.Yang, C.-C. Tsai, P.-L. Chiang, C.-Y. Pan, and Y.-T. Chen
Proc. Natl. Acad. Sci. U. S. A., 107, 1047-1052 (2010)
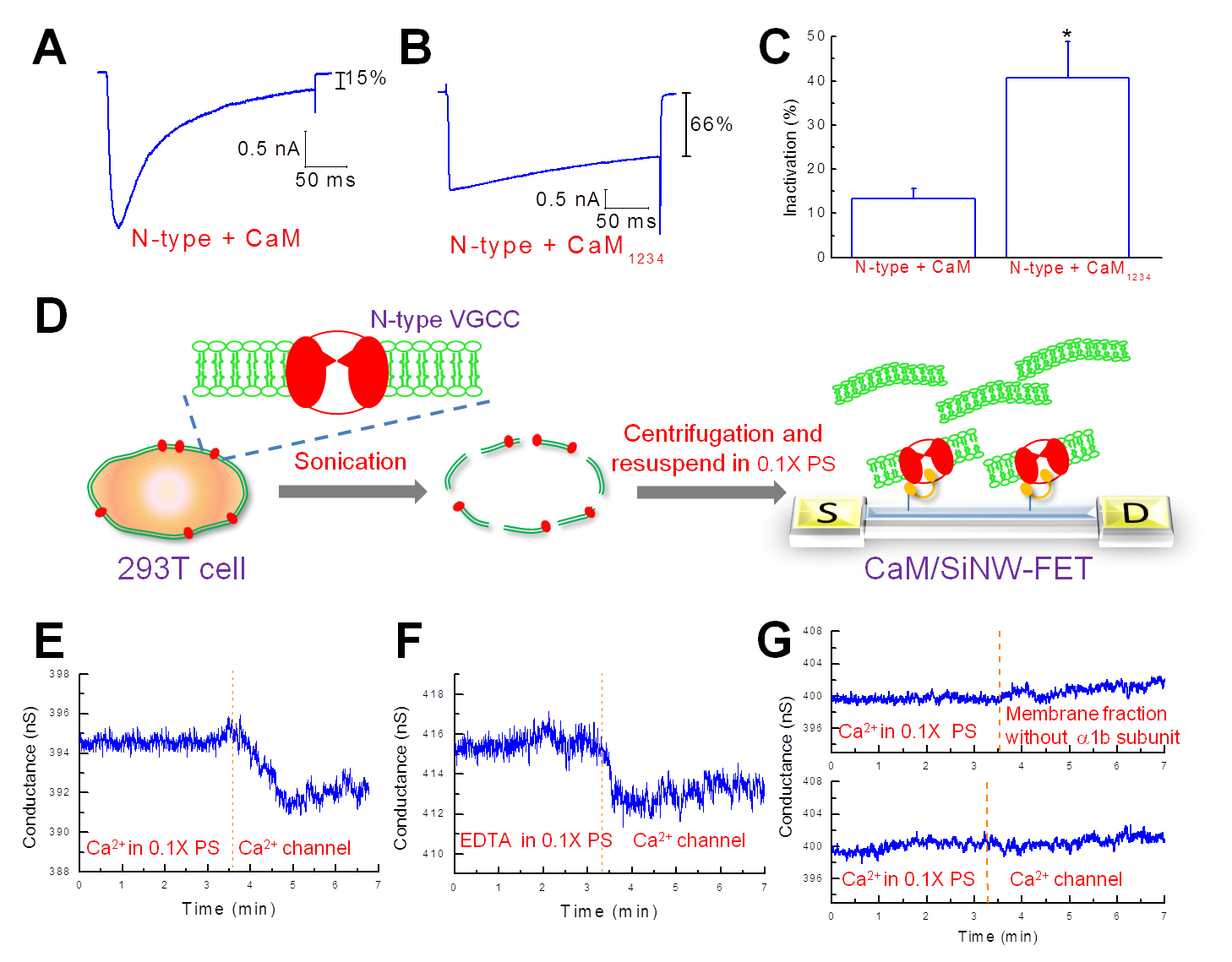
In this study, we describe a highly sensitive and reusable silicon nanowire field-effect transistor for the detection of protein-protein interactions. This reusable device was made possible by the reversible association of glutathione S-transferase-tagged calmodulin with a glutathione modified transistor. The calmodulin-modified transistor exhibited selective electrical responses to Ca2+ (≥1 μM) and purified cardiac troponin I (∼7 nM); the change in conductivity displayed a linear dependence on the concentration of troponin I in a range from 10 nM to 1 μM. These results are consistent with the previously reported concentration range in which the dissociation constant for the troponin I-calmodulin complex was determined. The minimum concentration of Ca2+ required to activate calmodulin was determined to be 1 μM. We have also successfully demonstrated that the N-type Ca2+ channels, expressed by cultured 293T cells, can be recognized specifically by the calmodulin-modified nanowire transistor. This sensitive nanowire transistor can serve as a high-throughput biosensor and can also substitute for immunoprecipitation methods used in the identification of interacting proteins.